What is TMS, and how can it help?
Safe & Effective Few Known Side Effects Non-Invasive No Anesthesia Outpatient Treatment Covered by Insurance (including Medicare)
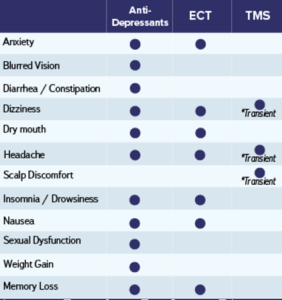
Magstim® TMS Therapy involves a series of repetitive, brief, and highly focused magnetic pulses, designed to stimulate brain cells. TMS is typically prescribed to adult patients when antidepressants have failed, or the side effects are intolerable. In comparison to antidepressants, Magstim® TMS Therapy is a non-invasive outpatient procedure with few known side effects1. Magstim® TMS Therapy systems offer versatility with a range of treatment protocols including a 3-minute treatment option.
What to Expect:
TMS therapy is a treatment that occurs five days a week for 6 weeks. Your doctor will determine the ideal stimulation intensity (dosage) and treatment protocol.
To determine the proper dosage, your doctor will perform a mapping called a ‘motor threshold assessment’. During this process, an electromagnetic coil will be placed against a part of your head called the motor cortex. A series of single pulses at different power levels will be delivered in order to find the specific intensity (dosage) to cause your thumb to twitch. The doctor will make calculations based on the number of thumb twitches and the intensity of the pulse to determine your dosage.
For treatment, the coil will be placed on the specific treatment location determined during the motor threshold assessment. Each treatment will last between 3 and 37.5 minutes depending on the clinical protocol prescribed by your physician.
TMS is a safe and well tolerated treatment with few known side effects. Clinical studies show that the most common side effects are mild to moderate scalp discomfort and mild headaches, both of which are short term7.
Side effects of different depression treatments